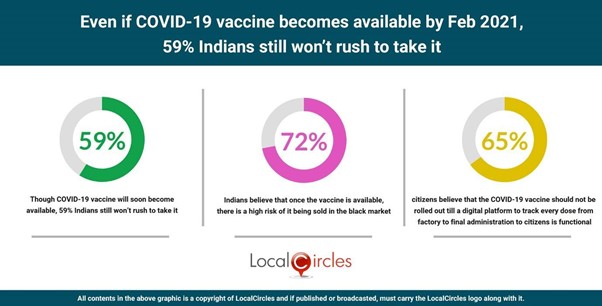
Despite an affordable COVID-19 vaccine now likely by Feb 2021, 59% Indians say they won’t rush to take it
- ● 22% Indians say they will get it as soon as it becomes available through any healthcare channel
- ● 72% Indians believe that there is a high risk of it being sold in the black market
- ● 65% Indians say the vaccine should be brought out only when the digital vaccine tracking platform is available
Even if COVID-19 vaccine becomes available by Feb 2021, 59% Indians still won’t rush to take it
December 2, 2020, New Delhi: The last few weeks have given hope to the world with three vaccine makers i.e. Pfizer, Moderna and Oxford-AstraZeneca releasing their efficacy data. While the Pfizer vaccine claimed efficacy of over 90 percent it requires storage temperature to be maintained at -70 degree centigrade, the Moderna vaccine in its latest release claimed an efficacy of 94.1% and requires storage temperatures of -20 degree centigrade.
The third vaccine, by Oxford-Astrazeneca is the one which holds promise for India as it can be stored at 2-8 degree centigrade temperature for up to 30 days and has claimed an average efficacy of 70%. Oxford-Astrazeneca have a partnership with Serum Institute of India for production and reports suggest that the vaccine could be available as early as February and at a MRP of 500-600 per dose. Oxford-Astrazeneca also said their vaccine was 90% effective when a half-dose was given before a full dose booster but two full doses only showed an efficacy of 62% confusing scientists, investors and citizens. Questions are being raised whether the approval process will go smoothly for Oxford-AstraZeneca as there are questions about transparency. Closer home in India, where Serum is conducting trials for the vaccine under name Covishield, a participant who undertook the trial has alleged the vaccine of causing him serious side-effects, both neurological and psychological filing for damages against Serum. Serum has dismissed the claims as “oblique pecuniary motive” maintaining that the participant’s suffering was independent of the vaccine trial he underwent and has sought damages for malicious allegations in excess of INR 100 crores (20 times the damages claimed by the participant). According to some experts, Serum should have made the adverse event public with some already calling this low level of transparency on the vaccine and it possibly leading to increase in distrust amongst citizens.
61% citizens of India had expressed hesitancy in getting the vaccine in a survey conducted between Oct 15-20th before any of the vaccines were announced. Now with all these developments in place and the possibility of an affordable vaccine by Feb 2021, LocalCircles decided to conduct another survey to understand if hesitancy levels have reduced. The survey received more than 25,000 responses from citizens across 262 districts of India. 69% respondents were men and 31% were women. 55% respondents were from tier 1, 24% from tier 2, and 21% were from tier 3, 4 and rural districts. Also, citizens’ reluctance to vaccinate themselves with the COVID-19 vaccine doses was also measured with previously conducted surveys on the subject to understand the change over time.
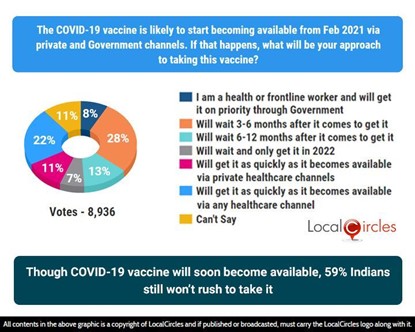
The first question in the survey asked, “The COVID-19 vaccine is likely to be available from February 2021 via private and Government channels. If that happens, what will be your approach to taking this vaccine?”. Responses from 8,936 citizens were received. Of which, 8% said “I am a health or frontline worker and will get it on priority through the Government”, 13% said “will get it as quickly as it becomes available via any healthcare channel”, 11% said “will get it as quickly as it becomes available via private healthcare channel”. Another 59% of respondents said that they won’t rush to take the COVID-19 vaccine even if it is available in the near future.
The perception of the majority of citizens sceptical on taking COVID-19 vaccine hasn’t changed much as compared to the result of the survey conducted in mid-October by LocalCircles. 61% of 8,312 voters at the time had said they are sceptical about COVID-19 vaccine and won't rush to take it in 2021 even if it is available.
Though COVID-19 vaccine will soon become available, 59% Indians still won’t rush to take it
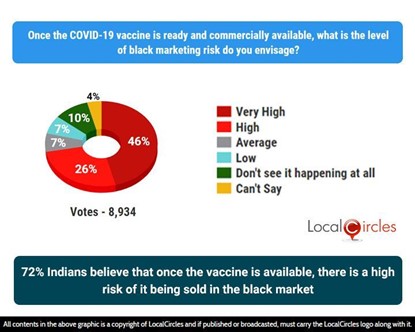
Since the inception of the pandemic, LocalCircles has been gathering citizens’ inputs on various issues in regards to the Covid outbreak. In July 2020, citizens via LocalCircles had alerted the Drug Controller General of India (DCGI) on blackmarketing of Remdesivir leading to immediate issuance of an order by DCGI to all state drug controllers asking them to up enforcement against the same. Many posts have been received from citizens in the last 3 weeks raising concerns of black marketing of vaccine. The second question asked citizens, “Once the COVID-19 vaccine is ready and commercially available, what is the level of black-marketing risk do you envisage?”. 8,934 responses were received. Notably, 72% Indians believe that once the vaccine is available, there is a high risk of it being sold in the market.
72% Indians believe that once the vaccine is available, there is a high risk of it being sold in the black market
In mid-July, after the Remdesivir black marketing escalation, LocalCircles on citizen recommendation wrote to Department of Pharmaceuticals and Ministry of Health & Family Welfare to implement serial number and bar code based tracking of vaccine from factory to administration to a patient such that there is no room for black-marketing. The same was acknowledged by Department of Pharmaceuticals which directed the National Pharmaceutical Pricing Authority (NPPA) to evaluate the same. In early October, the Government announced the implementation of a digital platform to track the administration of the COVID-19 vaccine, movement, storage and distribution to individual beneficiaries. However the timeline of this platform’s availability is still unclear.
65% citizens believe that the COVID-19 vaccine should not be rolled out till a digital platform to track every dose from factory to final administration to citizens is functional
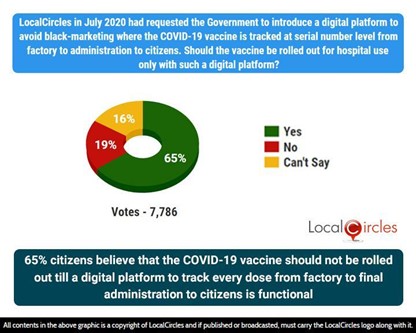
The third question asked citizens, “Should the vaccine be rolled out for hospitals only after a digital platform is introduced with the use of a unique serial number to track the supply of COVID-19 vaccine from factory to administration to citizens?”. Of 7,786 responses, 65% of citizens believe that the COVID-19 vaccine should not be rolled out till a digital platform to track every dose from factory to final administration to citizens is functional.
With 65% citizens believing that the vaccine should not be rolled for commercial or hospital use till the digital vaccine tracking platform is ready indicates that people of India believe that there is a high risk that the vaccine without a working digital platform could get in the wrong hands leading to black-marketing scenarios and even counterfeiting. The digital vaccine tracking platform, would reportedly need to track the supply of close to 1.7 billion Covid-19 vaccine doses needed to immunise most of India’s adult population – a mammoth task than any in the history, keeping in mind the country’s experience with large immunisation campaigns. The Prime Minister has asked all States to set up steering committees and task forces, down to block level preparation for Covid-19 vaccine disbursal so India is prepared when the vaccine does become ready.
LocalCircles will share the findings of this report with relevant Government stakeholders so the public concerns and feedback finds place in the strategy and implementation of COVID-19 vaccine roll out.
Survey Demographics
25,000+ responses were received from citizens across 262 districts of India. 69% respondents were men while 31% respondents were women. 55% respondents were from tier 1, 24% from tier 2 and 21% respondents were from tier 3, 4 and rural districts. The survey was conducted via LocalCircles platform and all participants are validated citizens who had to be registered with LocalCircles to participate in this survey.
About LocalCircles
LocalCircles, India’s leading Community Social Media platform enables citizens and small businesses to escalate issues for policy and enforcement interventions and enables Government to make policies that are citizen and small business centric. LocalCircles is also India’s # 1 pollster on issues of governance, public and consumer interest. More about LocalCircles can be found on https://www.localcircles.com
For more queries - media@localcircles.com, +91-8585909866
All content in this report is a copyright of LocalCircles. Any reproduction or redistribution of the graphics or the data therein requires the LocalCircles logo to be carried along with it. In case any violation is observed LocalCircles reserves the right to take legal action.


